Contents
ENZYME IMMOBILIZATION
Many enzymes are produced by microorganisms on a large scale and their production is cost effective even if they are used only once. However, more enzymes are such that they affect the cost of their production and may not be economical, if not reused. This necessity of reuse of enzymes led to the development of immobilization techniques, which basically involve process conversion of water-soluble enzyme protein into a solid form of catalyst using several methods. It is only possible to immobilize microbial cells by similar techniques. Immobilization is the imprisonment or arresting of an enzyme in a distinct phase that allows exchange with, but is separated from the bulk phase in which the substrate, inhibitor or effector molecules are dispersed and monitored. The first commercial application of immobilized enzyme technology was reported from Japan in 1969 with the use of A. oryzae amino acylase for the industrial production of L-amino acids. Consequently, pilot plant processes were reported for production of 6-aminopenicillanic acid (6-APA) from penicillin G and for conversion of glucose to fructose by immobilized glucose isomerase.
Advantages of Immobilized Enzymes
The advantages of immobilized enzymes are as follows:
- They can be reused.
- The production is less labour intensive.
- The continuous use is possible.
- There are less chances of contamination with their use.
- The reaction time required is minimized.
- The saving in capital cost is possible.
- High enzyme: substrate ratio can be achieved.
- They are more stable as compared to conventional means.
- Improved process control is a reality.
The first immobilized enzymes to be scaled up for industrial production are immobilized amino acid acylase, penicillin G acylase and glucose isomerase. Some other industrially important enzymesproduced by this technique are aspartase, esterase and nitrilase.
Techniques of Enzyme Immobilization
The different techniques of enzyme immobilization are
- physical immobilization,
- chemical immobilization,
- adsorption,
- covalent bonding,
- entrapment,
- copolymerization or crosslinking and
- encapsulation.
Physical Immobilization
It forms no covalent bonds between the enzymes and the supporting matrix. Earlier approaches include the adsorption of the enzyme onto animal charcoal or alumina, but current advancements make use of ionic adsorption technique onto ion-exchange resins, especially those of the sephadex-type and controlled-pore glass.
Advantages
- Simplicity, general applicability and high yield.
- It confers ability to replace the immobilized enzyme when its catalytic activity is decreased below an acceptable level.
Limitations
- It needs to control the working conditions for the use of immobilized enzyme to prevent its desorption.
- Products of enzyme entrapment in liposomes (artificially produced concentric spheres of phospholipids bilayers), polyacrylamide and agarose suffer from poor flow properties, inefficiency and progressive leaching of the enzyme.
- Desorption of the protein resulting from changes in temperature, ionic strength and hydrogen ion concentration.
Chemical Immobilization
The covalent bond formation between the enzyme and the matrix procedures are identical to those used in affinity chromatography. A loss of activity would be observed if attachment involves amino acid residues at the active site of the enzyme. The matrix may be polysaccharide or polymers (nylon or inorganic carriers, such as glass and titanium dioxide).
Applications
The first reported application is the production of L-amino acids from immobilized aminocylase. The other applications are as follows:
- Immobilized glucose isomerase in food industry for production of high fructose syrup a fructose is 16 times sweeter than glucose.
- Production of a chiral molecule for pharmaceutical and research purposes.
- Immobilized enzymes have an increasing number of analytical applications, especially in clinical situation, where they differ in the potential for fast, sensitive and accurate determinations of analytes, such as urea and blood glucose.
- A number of immobilized enzymes are also becoming important parts of a variety of biosensors.
- Combination of immobilized enzymes with high specificity and sensitivity, and use in electroanalytical chemistry is possible.
- Another recent development involves the quantification of enzymes, substrates and other analytes in samples of clinical importance. For this, the reagents including the enzyme are impregnated onto an immobile structure. On addition of a drop of test sample to the matrix, the analyte in the sample triggers an enzyme reaction, resulting in the formation of coloured product. The intensity of colour can be measured with the help of reflectance spectroscopy.
- It offers an advantage of accurate analysis without the need of sample preparation.
Adsorption
In this technique, an enzyme is immobilized by bonding either to the external or internal surface of a carrier or support, e.g. mineral support (aluminium oxide, clay), organic support (starch). modified sapharose and ion-exchange resins. In this process, bonds of low energy, such as ionic interactions, hydrogen bonds, van der Waals forces, etc., are involved. If the enzyme is immobilized externally, the carrier particle size must be very small (500 Å to 1 mm) in order to achieve an appreciable surface of bonding. In this technique of external immobilization, no pore diffusion limitations are encountered. The enzyme immobilized on an internal surface is protected from abrasion, inhibitory bulk solutions and microbial attack, and a more stable and active enzyme system can be achieved. The pore diameters of carriers may be optimized for internal surface immobilization.
Immobilization by adsorption is achieved by the following procedures:
- Static process: The solution containing the enzyme is allowed to contact the carrier without stirring for immobilizing the enzyme.
- Dynamic batch process: In this process, the carrier is placed into the enzyme solution and mixed by stirring or agitated continuously in a shaker.
- Reactor loading process: The carrier is placed into the reactor, then the enzyme solution is transferred to the reactor and the carrier is loaded in a dynamic environment by agitating the carrier and enzyme solution together.
- Electroposition process: The carrier is placed proximal to one of the electrodes in an enzyme bath, and the current is put on. The enzyme migrates to the carrier, and it is deposited on the surface and immobilization takes place.
- Covalent bonding: It is formed between the chemical groups of enzyme and the surface of carrier. Covalent bonding is utilized under a broad range of pH, ionic strength and other variable conditions. Immobilization steps are attachment of coupling agent followed by an activation process or attachment of a functional group, and finally attachment of the enzyme. Different types of carriers are used in this technique of immobilization, such as carbohydrates. proteins, inorganic carriers, amine-bearing carriers, etc. Covalent attachment may be directed to a specific group (e.g. amine hydroxyl, tyrosyl, etc.) on the surface of the enzyme. Hydroxyl and amino groups are the main groups of the enzyme with which bonds are formed. Different methods of covalent bonding are as follows:
- Diazotization: It refers to bonding between the amino group of the support (e.g. aminobensyl cellulose, aminosilanized porous glass, amino derivatives) and a tyrosyl or histidyl group of the enzyme.
- Formation of peptide bond: It refers to formation of bond between the amino or carboxyl group of the support and amino or carboxy group of the enzyme.
- Group activation: Cyanogen bromide is used for bonding to a support containing glycol group, i.e. cellulose, syphadex, sepharose, etc.
- Polyfunctional reagents: It involves use of a bifunctional or multifunctional reagent, such as glutaraldehyde, that forms bonding between the amino group of the support and amino group of the enzyme.
- The major problem encountered with covalent bonding is that the enzyme may be inactivated due to changes in conformation while undergoing reaction at active site. However, this problem may be overcome through immobilization in presence of enzyme’s substrate or a competitive inhibitor or protease. The most commonly used activated polymers are celluloses or polyacrylamides onto which diazo, carbodiimide or azide groups are incorporated.
- Entrapment: The enzymes can be physically entrapped inside a matrix (support) of water soluble polymer (e.g., polyacrylamide-type gels) and naturally derived gels (e.g. cellulose triacetate, agar, gelatin, carrageenan, alginate, etc.). The pore size of matrix is adjusted to prevent loss of enzyme from the matrix due to excessive diffusion. There is possibility of leakage of low molecular weight enzymes from the gel. Agar and carrageenan have large pore sizes (<10 µm). Different methods for enzyme entrapment used are as follows:
- Gel entrapment: In this process, the enzyme is entrapped in gels.
- Fibre entrapment: The enzyme is entrapped in fibre format.
- Microcapsule entrapment: The enzymes are entrapped in microcapsules of monomer mixtures, such as polyamine and polybasic chloride, polyphenol and polyisocyanate. The entrapment of enzymes has been widely used for sensing application, but not much success has been achieved with industrial process.
- Crosslinking or copolymerization: It is widely used in commercial applications. It is characterized by covalent bonding between various molecules of an enzyme via a polyfunctional reagent, such as glutaraldehyde, diazonium salt, hexamethylene disocyanate, and N-N’ ethylene bismaleimide. The polyfunctional reagent is capable of denaturing the enzyme. This technique is simple and economical, but not often used with pure proteins, because· it produces very little of immobilized enzyme that has very high intrinsic activity.
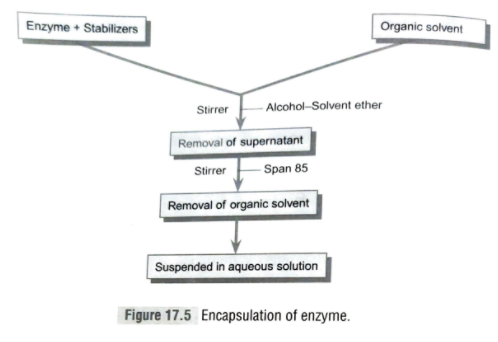
- Encapsulation: Chent has described the method of encapsulation in 1977. It is the process of enclosing a droplet of solution of enzyme in a semipermeable membrane capsule made up of cellulose nitrate and nylon. The method of encapsulation is simple and economical. Although, the catalyst is very effectively retained within the capsule, the effectiveness of this technique largely depends on stability of enzyme. It has limited applications to medical sciences. In this method, a large quantity of enzyme is immobilized, but main limitation of this technique is that only small substrate molecule is utilized with the intact membrane.
Cell Immobilization
This technique is how well developed and successfully used for industrial-scale production. In cell immobilization technology, the enzymes are active and stable for a long period of time.
The methods of cell immobilization is same as described for enzyme immobilization, involving the processes of adsorption, covalent bonding, cell-to-cell crosslinking, encapsulation and entrapment in polymeric network. Since a long time, adsorption of cells to preformed carrier has been undertaken. The cell attachment to the surface of preformed carrier is done by covalent bonding. The woodchips are used as carrier for Acetobacter in production of vinegar since 1823. Preformed carrier of specific choice is used.

APPLICATIONS OF ENZYMES
Enzymes in general are limited in the number of reactions they have evolved to catalyse and also by their lack of stability in organic solvents and at high temperatures. Consequently, protein engineering is an active area of research and involves attempts to create new enzymes with novel properties, either through rational design or in vitro evolution.
The enzymes and cells have wide spectrum of applications that can be grouped into four broad categories: (1) therapeutic, (2) analytical, (3) manipulative and (4) industrial.
- Therapeutic applications: Some inborn errors of metabolism occur due to missing of enzyme where specific genes are introduced to encode specific missing enzymes. Certain diseases are treated by administering the appropriate enzyme. For example, virilization of a disease developed due to loss of hydroxylase enzyme from adrenal cortex and introduction of hydroxyl group (-OH) on 21-carbon in ring structure of steroid hormone. The missing enzyme synthesizes aldosterone in excess leading to masculinization of female baby and precocious sexual activity in males within 5-7 years. Similarly, treatment of leukaemia (a disease in which leukaemic cells require exogenous asparagines for their growth) is possible by administering asparaginase of bacterial origin.
- Analytical applications: The enzymes are used in kinetic analysis. Endpoint analysis refers to total conversion of substrates into products in a few minutes in the presence of enzymes, whereas kinetic analysis involves the rate of reaction and substrate/product concentration. The analysis of antibodies and immunoglobins, necessary for human use, is also possible with enzymes. The usable enzymes are alkaline phosphatase, ß-galactosidase, lactamase, etc. Another important utility of enzyme is in biosensor. It is a device of biologically active material displaying characteristic specificity with chemical or electronic sensor to convert a biological compound into an electronic signal. A simple carbon electrode, an ion sensitive electrode, oxygen electrode or a photocell may be used as a biosensor.
- Manipulative applications: A variety of enzymes isolated from different sources are used in genetic engineering as one of the biological tools.
- Industrial applications: The industrial uses of enzymes may be broadly categorized as follows:
- Detergent industry: The stains on cloth can be easily removed by adding proteolytic enzyme to the detergent. The enzyme attacks on peptide bonds and therefore, dissolves protein. The alkaline serine protease obtained from B. licheniformis is commonly used in manufacture of detergent. In addition, the serine protease of Bacillus amyloliquefaciens that contains alpha-amylase is also extensively used.
- Starch industry: Currently, various enzymatic processes are successfully applied for different products. Glucose isomerase is an important enzyme used commercially in conversion of glucose to fructose via isomerization. Fructose is used for fructose syrup preparation. The enzyme glucose isomerase is widely used in production of fructose syrup. The reaction mixture at the end contains 42% fructose, 52% glucose and 6% dextrins. The mixture is sweeter than glucose and as sweet as sucrose. The technique has been developed to obtain 55% fructose concentration in syrup.
- Rubber industry: Catalase is used to generate oxygen from peroxide to convert latex into foam rubber.
- Photographic industry: Protease (ficin) dissolves gelatin of scrap film allowing recovery of its silver content.
- Dairy industry: For a long time, calf rennet has been used in dairy industry. In recent years, calf rennets are replaced by microbial rennets (e.g. Mucor michei) that are acid aspartate proteases. Lactase produced by Bacillus stearothermophilus is used for hydrolysis of lactose in milk. The enzyme lipase is used for flavour development in cheeses.
- Brewing industry: The enzymes commonly used in brewing industry are alpha-amylase, ß-glucanase and protease that are required for malt in substitution of barley. The biological source for these enzymes is B. amyloliquefaciens. The neutral protease helps in the inhibition of alkaline protease by an inhibitor, ß-glucanase that takes care of filtration problems due to poor quality of malt.
- Wine industry: The pectic enzymes such as pectin trans-eliminase (PTE), polymethyl galacturonase (PMG), polygalacturonase (PG), pectinesterase (PE), etc. are used in wine industry for high yields with improved quality. The peptic enzymes give good result when combined with other enzymes, e.g. protease glucoamylase.
- Pharmaceutical Industry: The enzyme penicillin ON acylase is widely used in the production of semisynthetic penicillins. All penicillins consist of an active ß-lactam ring, i.e. 6-aminopenicillanic acid (6-APA) group combined with different side chains (R group). The enzyme penicillin ON acylase removes ON group resulting in the separation of 6-APA and R groups. Finally, new synthetic side chains are coupled with 6-APA to synthesize new semisynthetic penicillins. Enzyme reaction is represented as follows:
The E. coli strains are the most explored and exploited ones for production of pencillin Gacylase. The biosynthesis of penicillin acylase in E. coli is controlled by alterations in concentrations of nutrients and culture conditions. Sudhakaran and Berkar (1989) reported the effect of growth substrate, inducers and regulators on formation of enzyme. The strain E. coli NCJM-2400 produced penicillin G acylase intracellularly when grown in nutrient broth containing phenylacetic acid (PAA). It was observed that PAA (20 mM) stimulated enzyme synthesis by 8-10 folds. Phosphate and yeast extract were found essential for both the growth and enzyme biosynthesis, whereas glucose, sorbitol, lactose, acetate and lactate (all 0.1%) catabolically repressed the enzyme formation.
¨C17C Penicillin V acylase occurs in fungal and actinomycetes sources. However, its activity has also been reported in many bacteria, such as Bacillus sphaericus, Erwinia aroideae and Pseudomonas acidovorans. Lowe et al. (1986) have identified a strain of Fusarium oxysporum that exhibited intracellular penicillin V acylase activity, which was induced by phenoxyacetic acid in culture. The enzyme was partially purified and concentrated from disrupted cells (cells hydrolysed with 5% penicillin V solution) by fractional precipitation with miscible solvents.
Paper industry: Amylases, xylanases, cellulases and ligninases degrade starch to lower viscosity, aiding sizing and coating paper. Xylanases reduce bleach required for decolourizing. Cellulases smoothen fibres, enhance water drainage and promote ink removal. Lipases reduce pitch and lignin-degrading enzymes remove lignin to soften paper.


