Contents
Aim: To prepare 6-Hydroxy nicotinic acid from Coumalic acid.
Requirements: Sulphuric acid, coumalic acid, methanol, sodium carbonate, hydrochloric acid, sodium hydroxide.
Theory: Methyl coumalate is formed from coumalic acid in the presence of methanol. 6-hydroxy nicotinic acid is formed by reaction between methyl coumalate and ammonia.
Reaction Involved:
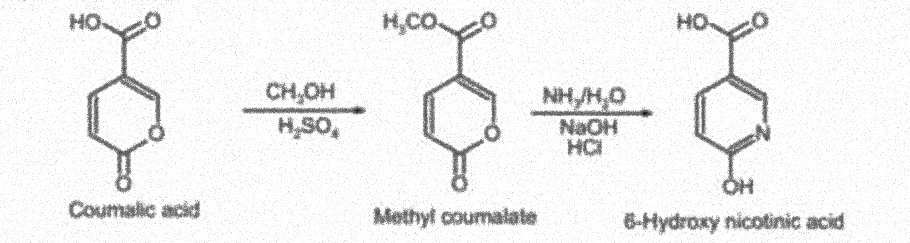
Procedure:
1. Preparation of Methyl coumalate:
- Place 100 ml of concentrated sulphuric acid in a 250 ml round bottom flask and add with swirling 35 g (0.224 mol) of finely powdered coumalic acid.
- The solution acquires a deep red-brown colour and the temperature tends to rise but should be kept below 30°C by cooling in an ice-water bath.
- Add 50 ml of methanol with continued swirling in small portions so that the temperature of the reaction mixture does not rise above 35°C.
- Heat the reaction mixture on a boiling water bath for 1.5 hours.
- Cool the mixture and pour slowly into 500 ml of ice-water slurry with stirring.
- Add about 150 g of sodium carbonate as slurry in mixture until the mixture is just neutral.
- If too much carbonate has been added, the supernatant liquid will acquire a red colour, in these circumstances add a few drops of sulphuric acid.
- The final solution containing a suspension of methyl coumalate and inorganic salts should be an orange colour.
- The precipitated ester is freed of inorganic salts by slurrying four times with 100 ml portions of cold water, filtered, and air-dried overnight.
- Filter off the methyl coumalate. The yield after air drying is 18 g (47%).
- • Although the product may be used for the next stage, a sample may be purified by vacuum sublimation; pure methyl coumalate is M.P. 74°C.
2. Preparation of 6-hydroxynicotinic acid:
- Place a mixture of 20 ml of ammonia solution (d = 0.88 g/ml) and 30 ml of water in a 250 ml beaker sited in an ice-water cooling bath on a magnetic stirrer unit.
- Insert a magnetic follower and add portion wise 18 g (0.105 mol) of methyl coumalate over a period of about 5 minutes, ensuring that the temperature is kept below 20°C.
- Stir the resulting dark red solution for a further 45 minutes at 20°C℃ and then add it to boiling aqueous sodium hydroxide prepared from 40 g of sodium hydroxide and 250 ml of water.
- Boil the reaction mixture for 5 minutes, cool to 10°C in an ice-water cooling bath and add concentrated hydrochloric acid with stirring to precipitate the product; the temperature of the solution during neutralization should not rise above 30°C.
- Allow the acidified solution to stand in the ice water bath for 1 hour and then collect the bright yellow crystalline solid by filtration.
- Wash the crystals with water and then air dry. The yield of 6-hydroxynicotinic acid, M.P. 299-300°C (decomp.) is 8.5 g (58%).
Results: The yield of obtained product was found to be _ gm and percentage yield was % and melting point was found to be °C.
Theoretical yield =
Practical yield =
Percentage of yield = ( Practical yield / Theoretical yield) * 100
Uses: It is an example of intermediate used in bulk drug industry. 6-Hydroxypyridine-3 carboxylic acid (6-Hydroxynicotinic acid) is used in the synthesis of molecularly imprinted polymer.
Caution: Glass ware should be clean and dry before being used.
